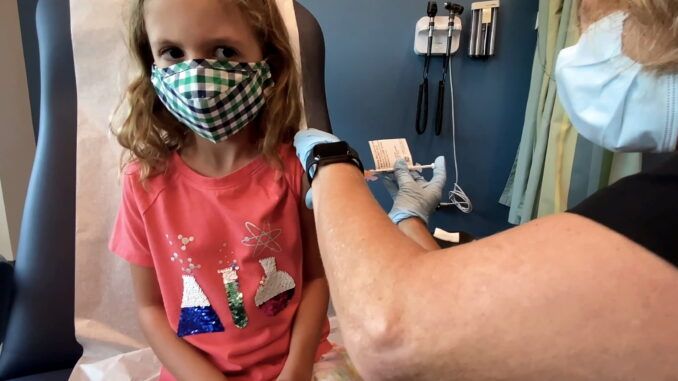
The US Food and Drug Administration (FDA) has approved Pfizer’s Covid-19 ‘vaccines’ for emergency use in children aged 5 to 11.
Fridays move potentially paves the way for millions of young children to soon be getting a covid shot.
According to a White House adviser, several US states have already put in their orders for the vaccine doses.

BYPASS THE CENSORS
Sign up to get unfiltered news delivered straight to your inbox.
You can unsubscribe any time. By subscribing you agree to our Terms of Use
The news comes as data, also released on Friday by the Centers for Disease Control and Prevention (CDC) shows that between Dec. 14, 2020, and Oct. 22, 2021, a total of 837,595 adverse events following COVID vaccines were reported to the Vaccine Adverse Event Reporting System (VAERS). The numbers include 17,619 deaths and 127,457 serious injuries.
Newsy Reports: Children’s hospitals, pediatricians and health centers are already preparing for getting the shots into arms. That includes letting parents know what to expect, especially for kids who don’t like needles.
“We have a buzzy that distracts your nerves so that you don’t know,” pediatrician Dr. Sharon Stoolman said. “It looks like a little bumblebee, but it kind of vibrates on the skin. We have cold spray.”
Now the move goes to the CDC. Its expert panel meets Tuesday. If Walensky signs off soon after, shots for 5- to 11-year-olds could start Wednesday, making them fully vaccinated by Christmas.

